Anti-HSP70 Monoclonal Antibody (Clone: BRM-22)
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 100 μg/vial |
| Isotype : | Mouse IgG1 |
| Purification : | Ascites |
| Content : | Mouse ascites fluid, 1.2% sodium acetate, 2mg BSA, with 0.01mg NaN3 as preservative. Reconstitute : Add 1ml of PBS buffer will yield a concentration of 100ug/ml. |
| Storage condition : | At -20˚C for one year. After reconstitution, at 4˚C for one month. It can also be aliquotted and stored frozen at -20˚C for a longer time. Avoid repeated freezing and thawing. |
Heat-shock proteins, or stress proteins, are expressed in response to heat shock and a variety of other stress stimuli including oxidative free radicals and toxic metal ions. Sargent et al. identified a duplicated HSP70 locus in the class III region of the major histocompatibility complex on 6p21.3. A duplicated locus encoding the major heat shock-induced protein HSP70 is located in the major histocompatibility complex(MHC) class III region 92 kilobases(kb) telomeric to the C2 gene. The 70-kd mammalian heat shock proteins are structurally and functionally related to the uncoating protein that releases clathrin triskelia from coated vesicles.
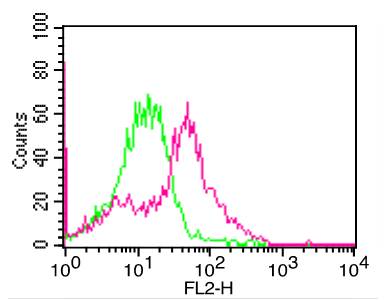
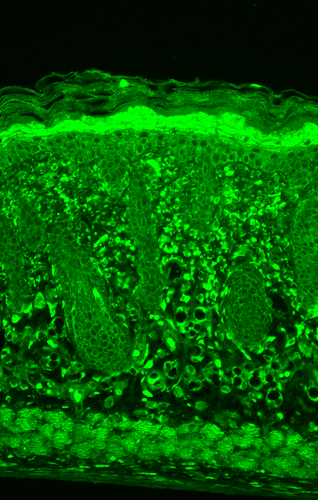
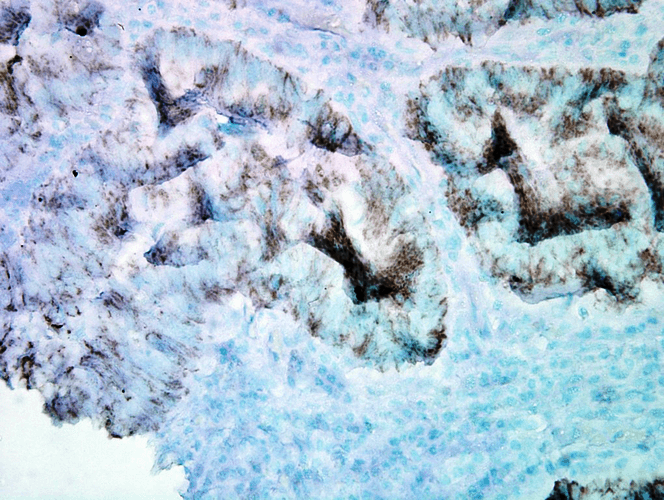
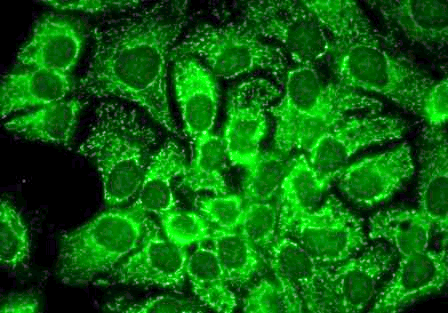
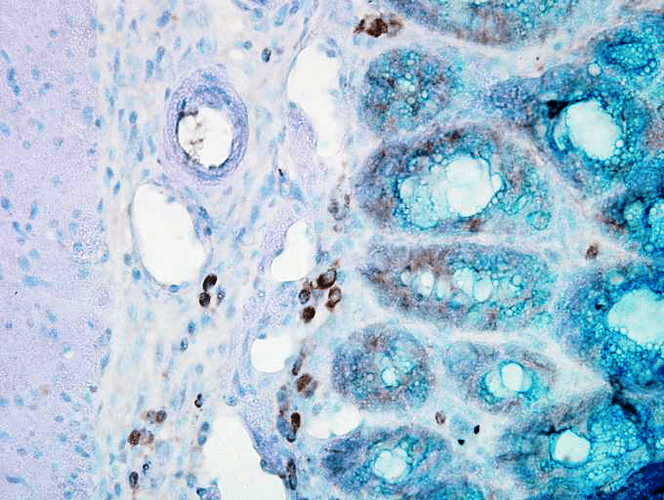
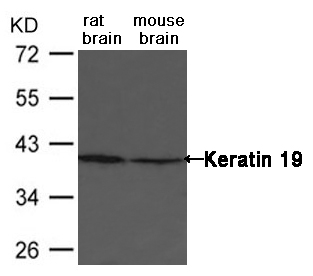
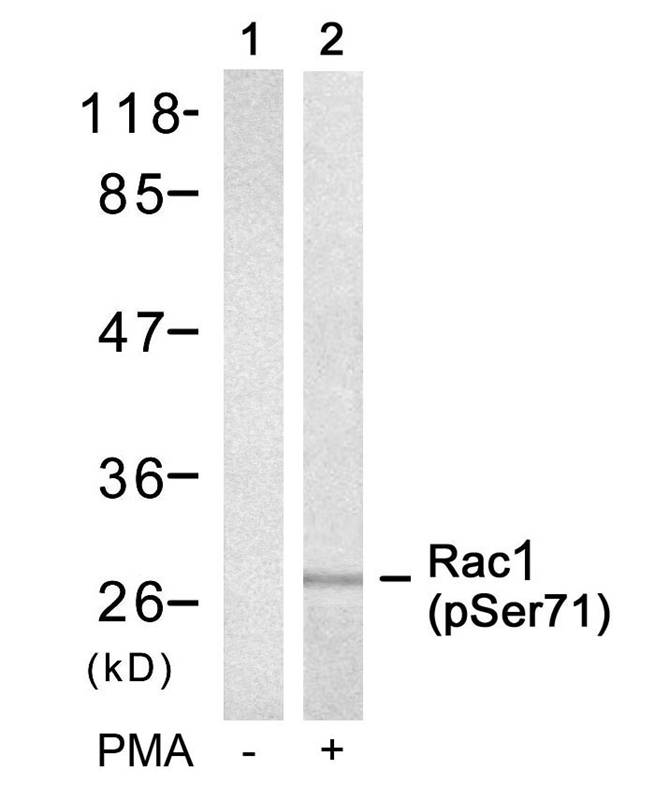
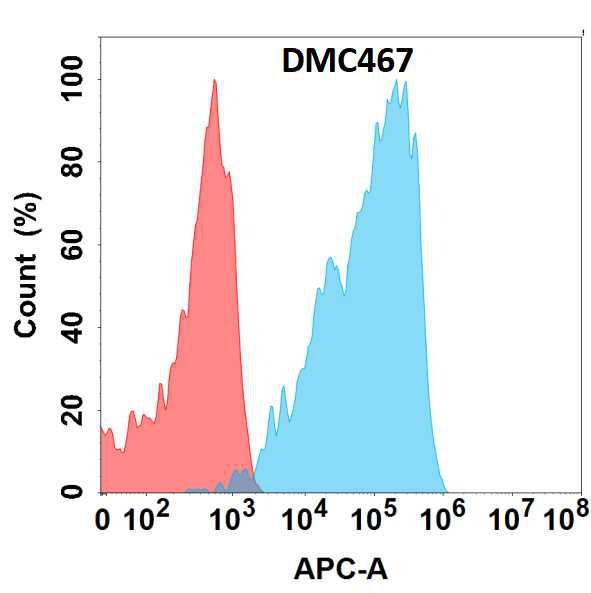
Western blot : 0.5μg/ml; Immunohistochemistry(Paraffin-embedded Section) : 0.5-1μg/ml; Immunohistochemistry(Frozen Section) : 0.5-1μg/ml
For Research Use Only. Not for use in diagnostic/therapeutics procedures.
| Subcellular location: | Cytoplasm, Cytoplasm |
| Post transnational modification: | In response to cellular stress, acetylated at Lys-77 by NA110 and then gradually deacetylated by HDAC4 at later stages. Acetylation enhances its chaperone activity and also determines whether it will function as a chaperone for protein refolding or degradation by controlling its binding to co-chaperones HOPX and STUB1. The acetylated form and the non-acetylated form bind to HOPX and STUB1 respectively. Acetylation also protects cells against various types of cellular stress. |
| Tissue Specificity: | Testis-specific. |
| BioGrid: | 200452. 39 interactions. |
|
There are currently no product reviews
|













.png)