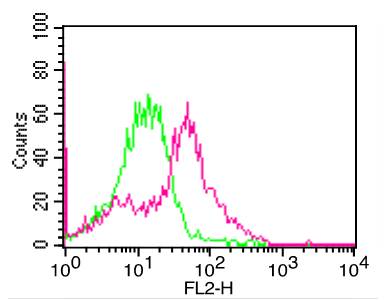
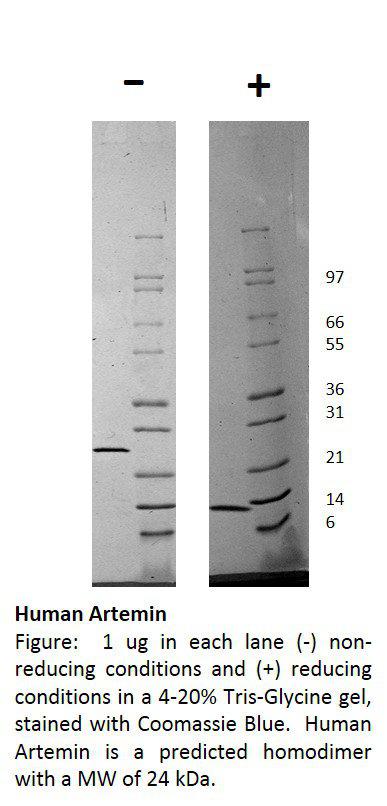
Influenza A Virus H3N2 Panama 2007/99
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 50 µg |
| Purification : | Greater than 90.0% as determined byAnalysis by SDS-PAGE. |
| Content : | The H3N2 A/Panama/2007/99 solution contains STE, 0.1% sodium azide (NaN3) and 0.005% thimerosal. |
| Storage condition : | A/Panama/2007/99 although stable 4°C for 4 weeks, should be stored desiccated below -18°C. Please prevent freeze-thaw cycles. |
Source : Allantoic fluid of 10 days old embryonated eggs, inoculated with influenza A virus, strain A/Panama/2007/99. The Influenza Virus was purified by Ultracentrifugation with 10-40 % sucrose gradient. H3N2 is a subtype of the influenza A virus. Its name derives from the forms of the two kinds of proteinson the surface of its coat, hemagglutinin(H) and neuraminidase(N). H3N2 exchanges genes for internal proteins with other influenza subtypes. H3N2 has tended to dominate in prevalence over H1N1, H1N2, and influenza B. H3N2 strain descended from H2N2 by antigenic shift, in which genes from multiple subtypes re-assorted to form a new virus. Both the H2N2 and H3N2 strains contained genes from avian influenza viruses.
Decription: Allantoic fluid of 10 days old embryonated eggs, inoculated with influenza A virus, strain A/Panama/2007/99. The Influenza Virus was purified by Ultracentrifugation with 10-40 % sucrose gradient.
Inactivation: Thimerosal and beta propiolactone treatmentThis product has been treated in a manner consistent with methods of inactivation. Generally accepted good laboratory practices appropriate to microbiological/viral safe handling practices and techniques are required at work.
|
There are currently no product reviews
|












.png)