Anti-Respiratory Syncytial Virus (Clone: RSV-14N4)
Shipping Info:
For estimated delivery dates, please contact us at [email protected]
| Amount : | 100 µg |
| Isotype : | Human IgG1κ |
| Purification : | ≥95% monomer by analytical SEC |
| Content : | ≥ 5.0 mg/ml. This recombinant monoclonal antibody is aseptically packaged and formulated in 0.01 M phosphate buffered saline (150 mM NaCl) PBS pH 7.2 - 7.4 with no carrier protein, potassium, calcium or preservatives added. |
| Storage condition : | Functional grade preclinical antibodies may be stored sterile as received at 2-8°C for up to one year. For longer term storage, aseptically aliquot in working volumes without diluting and store at ≥ -70°C. Avoid Repeated Freeze Thaw Cycles. |
| Alternative Name : | RSV, Orthopneumovirus, |
| Immunogen Information : | Human donors targeting the postfusion RSV F protein using human hybridoma technology |
Reactivity Species : Respiratory Syncytial⋅Virus
Expression Host : HEK-293
Endotoxin Level : ≤ 1.0 EU/mg as determined by the LAL method
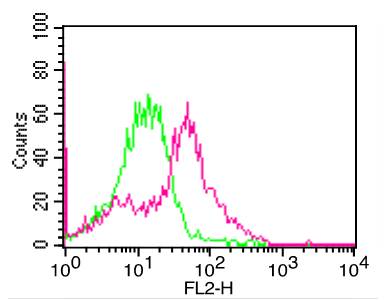
Specificity : RSV-14N4 activity is directed against antigenic site II of the RSV fusion (F) protein. RSV-14N4 readily competes with clone RSV-12I1 on post-fusion F, but the competition is less pronounced on prefusion F.
A plaque reduction neutralization assay showed RSV-14N4 is capable of neutralizing RSV strain A2. By ELISA RSV-14N4 binds to both prefusion and post-fusion F proteins with equal affinity. Competition-binding studies showed that RSV-14N4 targets antigenic site II, which is the target of palivizumab, an antiviral monoclonal antibody used as a prophylactic treatment. Saturation alanine scanning mutagenesis identified residues Asp263, Ile266, Asp269, and Lys271 as critical for 14N4 binding. Binding to antigenic site II was confirmed by x-ray crystallography and electron microscopy as well as by binding to scaffolded epitopes containing site II.
|
There are currently no product reviews
|














.png)